I am not a doctor nor am I in the medical field. But, during times like this, when other countries are having success with new or old drugs and saving their citizens’ lives, why wouldn’t you be doing the same thing? Why isn’t there constant communication between our government and the UK to share successes and failures in the treatment of covid-19?
I shake my head in disbelief as I read story after story of successful antibody treatments that are saving thousands of lives every day in other countries, while we continue to give American’s treatments with our wonder drug Remdesivir (Death-is-near).
Clearly, the failure here starts at the top with Fauci and spills over onto the Biden administration. And at this point, you have to stop and ask yourself if the administering of this drug and the increased deaths over 2020 are on purpose?
Despite knowing that this drug was taken off the Ebola trial because it killed 55% of the people that took it, the New England Journal of Medicine released a study yesterday that now says that Remdesivir is a “wonder drug” and saved 87% of the “unvaccinated” patients that they treated with it. Right! Buyer beware! How did this drug go from killing 55% of the patients that took it to saving 87% of the patients that took it? It didn’t.
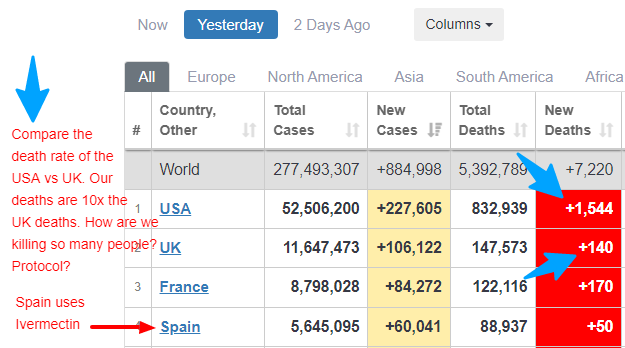
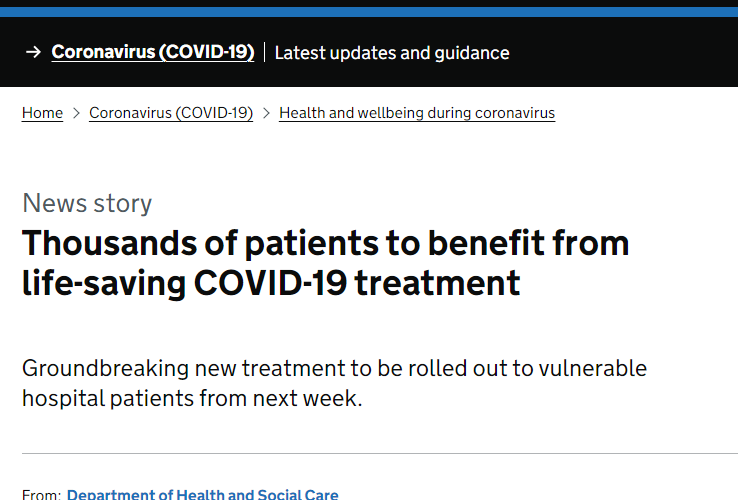
Below it an article about the new treatment being used in the UK with great success. I am also attaching a screenshot of the death numbers in the UK vs the US during the Omicron variant flareup. Look at how few deaths they have compared to the US! What are we doing! It’s clearly time to replace Fauci and the whole medical staff directing this ship.
>>>>>>>>ARTICLE COPIED FROM UK – LINK BELOW>>>>>>>>>>>>
Thousands of vulnerable NHS patients in hospital due to COVID-19 are set to benefit from a groundbreaking new antibody treatment, the government has announced today (17 September 2021).
Ronapreve, a combination of 2 monoclonal antibodies, will be targeted initially at those in hospital who have not mounted an antibody response against COVID-19.
This includes people who are immunocompromised, for example those with certain cancers or autoimmune diseases, and therefore have difficulty building up an antibody response to the virus, either through being exposed to COVID-19 or from vaccination.
The government has taken action to secure supply of the new therapeutic for NHS patients across the 4 nations, buying enough to treat eligible patients in hospital from next week. Guidance will shortly be going out to clinicians so they can begin prescribing the treatment as soon as possible.
Health and Social Care Secretary Sajid Javid said:
We have secured a brand new treatment for our most vulnerable patients in hospitals across the UK and I am thrilled it will be saving lives from as early as next week.
The UK is leading the world in identifying and rolling out life-saving medicines, particularly for COVID-19, and we will continue our vital work to find the best treatments available to save lives and protect the NHS.
Ronapreve is the first neutralising antibody medicine specifically designed to treat COVID-19 to be authorised by the Medicines and Healthcare products Regulatory Agency (MHRA) for use in the UK.
It will be used to treat patients without antibodies to SARS CoV-2 who are either aged 50 and over, or are aged 12 to 49 and are considered to be immunocompromised.
Antibody testing will first be used to determine whether patients are seronegative, meaning those who do not have an adequate existing antibody response, and will therefore receive the treatment. The treatment antibodies – casirivimab and imdevimab – will then be administered to patients through a drip and work by binding to the virus’s spike protein, stopping it from being able to infect the body’s cells.
The UK’s world-renowned vaccination programme also continues to provide protection to tens of millions of people across the country, and has so far saved 112,300 lives, prevented 230,800 hospitalisations and stopped over 24 million infections in England alone.
Since the beginning of the pandemic, the UK has proven itself to be a world-leader in identifying and rolling out effective treatments for COVID-19 – including the world’s first treatment, dexamethasone, which has since saved at least 22,000 lives in the UK so far and an estimated million worldwide.
The NHS has also rolled out monoclonal immunomodulatory antibody treatments tocilizumab and sarilumab, following clinical trial results from the government-funded REMAP-CAP trial. The treatments were found to reduce the relative risk of death by 24%, when administered to patients within 24 hours of entering intensive care.
Earlier this year, the government also brought together a new Antivirals Taskforce to supercharge the search for new treatments for patients who are exposed to COVID-19 to stop the infection spreading and speed up recovery time.
The UK’s leading research infrastructure and life sciences sector makes it the ideal base for the brightest of global innovators to research and progress cutting-edge treatments for COVID-19 through the clinical trials process here in the UK.
Paul McManus, COVID-19 Lead at Roche Products Ltd, said:
“Over the last 18 months, our goal has been to do everything we can to minimise the impact of the pandemic on those affected and the brilliant people who work tirelessly to treat and care for them. Ronapreve is the first dedicated medicine developed for COVID-19 to receive marketing authorisation from the MHRA, representing a significant milestone in how the NHS is able to fight this disease.
This is just another step in our journey to overcome COVID-19, and we will continue to collaborate with partners to identify and investigate multiple options that may help different groups of patients. Together with Regeneron, we’re grateful for the collaboration of the Vaccine Taskforce and NHS England in helping to bring this important antibody cocktail to treat and prevent acute COVID-19 across the UK.”